Tegeticula
Yucca moths
Olle Pellmyr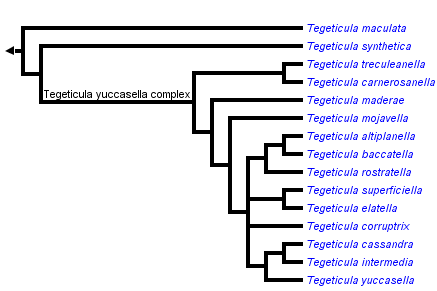

This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxBased on Pellmyr et al (1996) and Pellmyr & Leebens-Mack (2000)
Introduction
Tegeticula is one of the two genera of true yucca moths, which offer one of the classical cases of coevolved obligate mutualism between species. Females use unique tentacles on their maxillary palps to actively collect and compact pollen from the flowers of their yucca hosts. Yucca pollen is very sticky, and it adheres (without aid of the tentacles) to the underside of her head. Once loaded, the female oviposits into yucca flowers, walking up to floral stigmas after oviposition and actively pollinating the flower with a small portion of her pollen batch. In so doing, she assures that lack of pollination will not cause the flower to abort. This is critical, because her progeny feed only on developing seeds. The larvae only feed on a modest proportion of the seeds, thus making it possible for this herbivore to actually be of net benefit to its host. Over evolutionary time, yucca moths and yuccas have evolved complete mutual dependence on each other (Riley 1892, Pellmyr et al 1996b).
A considerable body of literature has accumulated on ecological and evolutionary aspects of the yucca-yucca moth interaction; a comprehensive reference list for the early literature is provided in Davis (1967), and much of the subsequent work is referenced in Powell (1992) and Pellmyr et al (1996). Recent empirical work has focused on the stability of obligate mutualism (Pellmyr and Huth 1994, Richter and Weis 1995), reversal of mutualism (Pellmyr et al 1996a), and moth behavioral plasticity in pollination and egg dispersal decisions (Addicott and Tyre 1995).
Recently the species traditionally called T. yuccasella has been identified as being a complex of at least 13 species, many of which have very distinctive biology. Among them are two non-pollinating species of 'cheater yucca moths'. The reader should bear in mind that many papers written before the recent revision that provide, e.g., measures of seed consumption, actually provide composite measurements of all coexisting species. This problem only applies to the yuccasella complex ("the white species").
Characteristics
Tegeticula shares one synapomorphy with Parategeticula:
- tentacle-like appendages arising ventrally between the first and second segments of the maxillary palps in the female (secondarily lost in some Tegeticula)
Two traits that distinguish Tegeticula from Parategeticula are:
- a sword-shaped ovipositor, used to cut into the host ovary
- three pairs of setae on the anal plate in the larva (two in Parategeticula)


Maxillary tentacles and pollen load of T. carnerosanella, a species in the Tegeticula yuccasella complex.
Life habits of immature stages
Tegeticula females oviposit into floral ovaries or developing fruits, and the grublike larvae feed of developing seeds. Usually white during the early instars, the larvae turn pink or to dark red when reaching later instars. The larva bores out through the fruit wall upon maturity, and creates a thick cocoon in the soil. It diapauses, sometimes for more than one year, and pupation apparently takes place a few weeks or less before adult emergence. The pupa is equipped with a frontal protrusion and large spines on the abdominal segments, which serve in burrowing up to the ground surface.


A last-instar larva of T. treculeanella, a member of the yuccasella complex.
Geographic distribution
The range of Tegeticula closely matches the extant distribution of their yucca hosts. This includes the extended ranges of yuccas in interior eastern North America, where European settlers introduced yuccas over the last 150 years. The northern limit of the genus is now in southern Alberta and Ontario in Canada. The southernmost records are from Tehuacan in Puebla, Mexico (Davis 1967).
Riley shipped larvae to Europe in an attempt to introduce yucca moths, but there is no report of success.
Discussion of Phylogenetic Relationships
The phylogenetic relationship between the three historically recognized species has only been considered in an analysis based on mitochondrial DNA sequence data (Pellmyr et al 1996). The topology is very strongly supported in that analysis. Phylogenetic resolution provided here is based on data from a paper to appear in the American Naturalist (Pellmyr & Leebens-Mack 2000). There is evidence of a rapid radiation event within this group, and relationships among the biologically diverse lineages is not yet clear.
References
Addicott, J.F and A.J. Tyre. 1995. Cheating in an obligate mutualism: How often do yucca moths benefit yuccas? Oikos 72:382-394.
Davis, D.R. 1967. A revision of the moths of the subfamily Prodoxinae (Lepidoptera: Incurvariidae). U.S. Nat. Hist. Mus., Bull. 255:1-170.
Frack, D.C. 1982. A systematic study of prodoxine moths (Adelidae: Prodoxinae) and their hosts (Agavaceae), with descriptions of the subfamilies of Adelidae (s. lat.). M.S. thesis, California State Polytechnic University, Pomona, CA.
Pellmyr, O. & C.J. Huth. 1994. Evolutionary stability of mutualism between yuccas and yucca moths. Nature 372:257-260.
Pellmyr, O. & J. Leebens-Mack. 2000. Reversal of mutualism as a mechanism of radiation in yucca moths. The American Naturalist 156:S62S76.
Pellmyr, O., J. Leebens-Mack & C.J. Huth. 1996a. Non-mutualistic yucca moths and their evolutionary consequences. Nature 380:155-156.
Pellmyr, O., J.N. Thompson, J. Brown & R.G. Harrison. 1996b. Evolution of pollination and mutualism in the yucca moth lineage. Amer. Nat. 148:827-847.
Powell, J.A. 1992. Interrelationships of yuccas and yucca moths. TREE 7:10-15.
Richter, K.S. and A.E. Weis. 1995. Differential abortion in the yucca. Nature 376:557-558.
Title Illustrations

| Scientific Name | Tegeticula yuccasella and Yucca filamentosa |
|---|---|
| Specimen Condition | Live Specimen |
| Behavior | Tegeticula yuccasella female in the process of actively pollinating a Yucca filamentosa flower. |
| Sex | Female |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1996

|
| Scientific Name | Tegeticula synthetica |
|---|---|
| Specimen Condition | Live Specimen |
| Behavior | resting in a host flower |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1996

|
About This Page

University of Idaho, Moscow, Idaho, USA
Correspondence regarding this page should be directed to Olle Pellmyr at
Page copyright © 1996
 Page: Tree of Life
Tegeticula. Yucca moths.
Authored by
Olle Pellmyr.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Tegeticula. Yucca moths.
Authored by
Olle Pellmyr.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 13 January 1997
Citing this page:
Pellmyr, Olle. 1997. Tegeticula. Yucca moths. Version 13 January 1997 (under construction). http://tolweb.org/Tegeticula/12419/1997.01.13 in The Tree of Life Web Project, http://tolweb.org/









 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site